Filters
Question type
A) The size of the activation energy tells us about the reaction mechanism.
B) The size of the activation energy tells us about the reaction rate.
C) A slow reaction has low activation energy.
D) A fast reaction has high activation energy.
E) A) and B)
F) A) and C)
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Question 32
Multiple Choice
How many transition states are present in the reaction in the energy diagram?

A) 0
B) 1
C) 2
D) 3
E) C) and D)
F) B) and D)
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Question 33
Multiple Choice
In which reaction is Keq > 1?
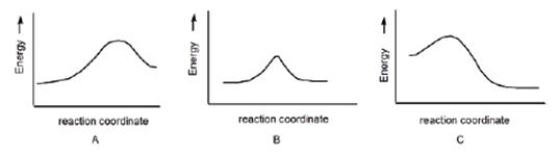
A) A
B) B
C) C
D) None of the above
E) B) and C)
E) B) and C)
Correct Answer

verified
Correct Answer
verified
Question 34
Multiple Choice
Which step would most likely have the largest energy of activation?
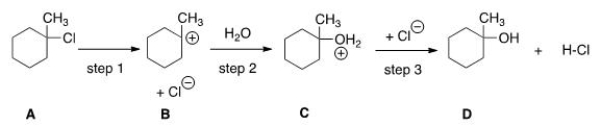
A) Step one
B) Step two
C) Step three
D) It cannot be determined from the information provided
E) A) and D)
F) B) and D)
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Question 35
Multiple Choice
What is the name given to the reaction species that lies at an energy minimum between steps on a reaction energy diagram?
A) Transition state
B) Activation energy
C) Reactive intermediate
D) Equilibrium product
E) None of the above
F) A) and B)
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 45 of 45
Related Exams